What is Raynaud’s Syndrome?
Raynaud’s Syndrome is a disorder of the blood vessels, usually in the fingers or toes, causing blood vessels to narrow when exposed to cold temperatures or during periods of stress. This narrowing, or vasoconstriction, leads to reduced blood perfusion to the affected sites.
This condition is also known as Raynaud’s Disease or Primary Raynaud’s when not accompanied by arterial obstruction. In cases where it is accompanied by arterial obstruction, it is referred to as Raynaud’s Phenomenon or Secondary Raynaud’s.
The terms “Raynaud’s syndrome,” “Raynaud’s disease,” “Raynaud’s phenomenon,” and “Raynaud’s disorder” are often used interchangeably.
When Raynaud’s occurs, blood cannot reach the surface of the skin, resulting in areas turning white or blue. In severe cases, this reduced blood flow can lead to sores or tissue death.

Raynaud’s Syndrome affects approximately 5% of the population, with a higher occurrence in females. It is more prevalent in regions with cold climates.
The risk factors for this condition include smoking and alcohol consumption.
To diagnose Raynaud’s Disease, physiological tests are typically performed. Such tests include measuring of photoplethysmography (PPG) waveforms with or without blood pressure in the fingers or toes, both at room temperature and after cold immersion.
Here is an example of a step-by-step guide for performing Raynaud’s Syndrome Test:
Remember, this step-by-step guide is a general outline for performing a Raynaud’s test. The specific protocol may differ among hospitals and institutions. Healthcare professionals should follow appropriate protocols based on their expertise and available resources.
Required for Raynaud’s Assessment

Optional Method for Raynaud’s Diagnosis
The Falcon/PRO system offers significant advantages for Raynaud’s diagnosis:
By utilizing the Falcon/PRO system’s advanced capabilities, healthcare professionals can benefit from a comprehensive and efficient diagnostic workflow. The system’s simultaneous measurement of multiple parameters and its ability to capture detailed data provide a more robust diagnosis and improved treatment planning.
During a Raynaud’s Syndrome test, several expected results and observations can be noted. First, during the cold immersion phase, the affected area may exhibit color changes, turning white or blue due to vasoconstriction and reduced blood flow. This discoloration serves as a characteristic sign of Raynaud’s Syndrome.
Additionally, the focus of the test is on the time it takes for the PPG waveforms to return to their pre-immersion amplitude levels after exposure to cold temperatures.
It is expected that the PPG waveforms will gradually recover and return to their baseline levels within approximately 10 minutes or so. This recovery time can vary depending on individual factors and the severity of the condition. A longer duration for the waveforms to return to pre-immersion levels may indicate the presence of a disease condition related to Raynaud’s Phenomenon.
Systolic blood pressure measurements can provide further insights into the diagnosis. In cases of secondary Raynaud’s, a decrease in systolic blood pressure compared to primary Raynaud’s is typically observed. This difference in blood pressure readings can help distinguish between primary and secondary forms of the condition.
To summarize, the expected results of a Raynaud’s Syndrome test include color changes in the affected area, gradual recovery of the PPG waveforms to their baseline levels within approximately 10 minutes, and potential variations in systolic blood pressure readings between primary and secondary Raynaud’s cases. These observations serve as important indicators for diagnosing and assessing the condition.
Wigley FM. Clinical practice. Raynaud’s Phenomenon. N Engl J Med. 2002;347(13):1001-1008.
Assessment of upper extremity arterial occlusive disease, Steven R. Talbot, in “Introduction to Vascular Ultrasonography”, Ed. Pellerito and Polak, Elsevier Health Sciences, 2012, Ch 15, pp 262-280
Herrick AL. Pathogenesis of Raynaud’s phenomenon. Rheumatology (Oxford). 2005 May;44(5):587-96
Three-Grade Classification of Photoplethysmography for Evaluating the Effects of Treatment in Raynaud Phenomenon, Shin-Seok Yang et al., Angiology 64(8) 609-613, 2012
Multifinger photoplethysmography and digital blood pressure measurement in patients with Raynaud’s phenomenon of the hand, J H Tordoir, L B Haeck, H Winterkamp, W Dekkers, J Vasc Surg, 1986 Mar;3(3):456-61.
The Thoracic Outlet Syndrome test (TOS) is an examination performed primarily to discriminate between the causes of patient symptoms due to compression at the thoracic outlet. The compression can affect the blood vessels or nerves, and the TOS test is conducted to determine whether the patient symptoms originate from vascular or neurogenic causes.
This diagnostic vascular TOS procedure tests for intermittent perfusion loss, particularly in the arms and hands. Either Doppler, PVR, or PPG sensors can detect normal resting waveforms in the digits or hands. Then, the patient is instructed through a sequence of positional maneuvers, which include the symptomatic position.
The examination tries to identify a position that significantly reduces perfusion to determine whether the symptoms originate from a vascular disorder. The examination is typically performed in parallel on both the right and left sides.

Gold Standard ABI Measurement Method

Secondary Method of ABI Assessment

High quality available in a variety of sizes
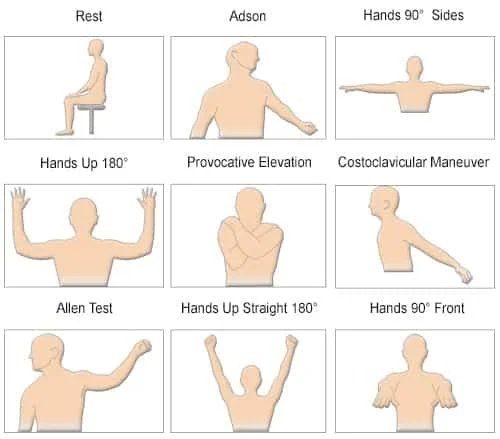
The Falcon is designed with a dedicated TOS protocol to allow a complete and effective diagnosis. The protocol guides both the examiner and the patient through a series of positional maneuvers that are visually presented with schematic pictures for clarity and to avoid mistakes. The range of positions includes maneuvers such as:
However, additional maneuvers can be added, and the protocol can be easily configured accordingly. The symptomatic position, which is probably the most important maneuver, is also included in the protocol. The examiner can use the Falcon camera to take a picture of the symptomatic position and document it in the protocol and subsequent report.
While standard TOS requires the use of PPG sensors (photoplethysmography sensors), the Falcon also supports a complete range of TOS testing capabilities, including Doppler and PVR measurements. All sensors can be placed for maximal efficiency, for example, on both the right and left digits, and these sites will be measured simultaneously. For even more effective evaluation, up to 5 PPG sensors and 10 PVR waveforms (for each finger) can be measured simultaneously.
The diagnosis of the TOS test is primarily visual. Still, the Falcon also provides a range of quantitative parameters, including PPG and PVR waveform amplitudes and the Doppler parameters range. These quantitative measurements help to support the qualitative findings.
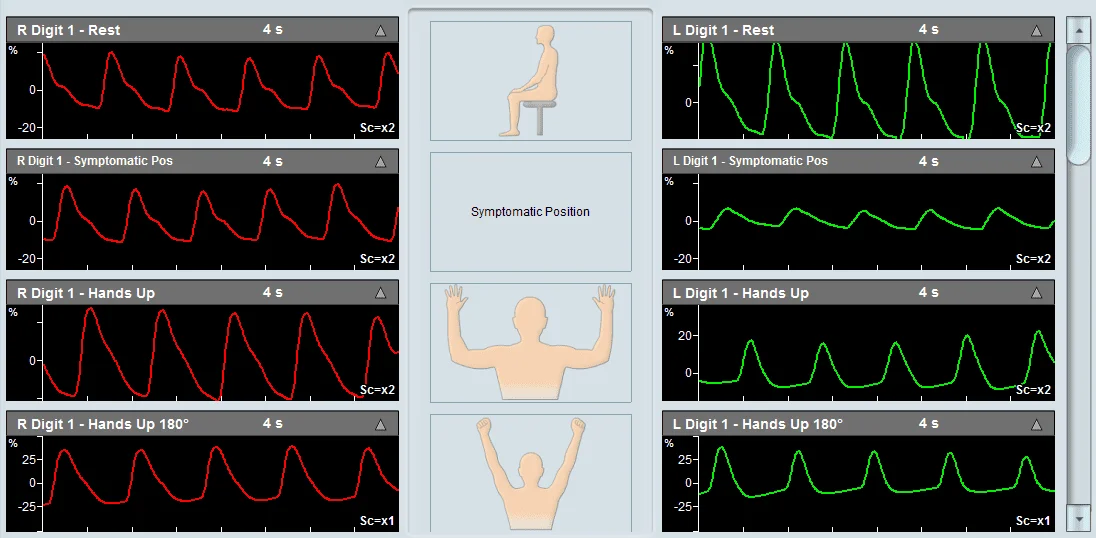
The diagnosis of the TOS test is primarily qualitative in nature. The focus is on viewing the measured waveforms on both the right and left sides for each positional maneuver and determining whether a specific position resulted in an abnormal decrease in amplitude or resulted in signal flattening. Such a case is indicative of a potential decrease in perfusion, suggesting a vascular source of TOS.
An example of a Thoracic Outlet Syndrome examination taken using Viasonix Falcon/PRO system.
Reporting standards of the Society for Vascular Surgery for thoracic outlet syndrome, Karl A. Illig et al., J Vasc Surg 2016;64:e23-e35.
Diagnosis and Treatment of Thoracic Outlet Syndrome, Editors Julie Ann Freischlag and Natalia O. Glebova, 2018 MDPI, Basel, Switzerland
The Palmar Arch Test (PAT), also known as Allen’s Test, is a specialty test performed to evaluate the patency of the arteries in the hand prior to radial or ulnar artery harvesting in artery bypass procedures or before the surgical creation of an upper extremity hemodialysis fistula or graft.
The PAT procedure provides a clinical assessment of the collateral circulation to the hand to determine if arterial harvesting is possible. The test is quite simple and involves sequential compression of the radial and ulnar arteries, and determining the physiological effect on the blood flow circulation in the hand. The blood flow assessment can be performed by different means, with the most popular method being with PPG sensors (Photoplethysmography).
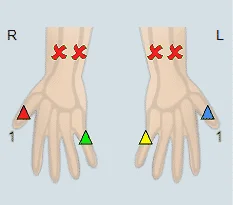
Additional diagnosis options include digit blood pressure measurements, PVR measurements (Pulse Volume Recording), and Doppler blood flow measurements. When using PPG sensors, it is preferred to use at least 2 PPG sensors, which are placed on the two extreme digits (digit 1 and digit 5). The Palmar Arch Test protocol begins by measuring the resting PPG waveforms, then measuring the waveforms after selected compressions (compression of the radial artery, ulnar artery, or both), and finally measuring the waveforms after the release of the selected compression. A similar protocol is applied when using digit pressure, Doppler, or PVR assessment.
Compression is typically performed by manually compressing the selected artery or arteries. This procedure allows the examiner to evaluate whether the result of the compression is impeded or complete loss of blood flow to parts of the hand. Thus, significant reduction or loss of blood flow to areas of the hand suggests that removing that particular artery may jeopardize hand performance if the artery is removed.

Secondary Method of ABI Assessment

High quality available in a variety of sizes
The Falcon has a dedicated Palmar Arch Test (PAT) protocol, which is designed to complete the assessment in guided procedures per the accepted guidelines. While the standard clinical assessment is with PPG sensors on the 2 extreme digits, the Falcon has the ability to place a PPG sensor on each of the 5 digits and perform the test simultaneously for all digits for optimal clinical assessment. The Falcon also supports digit blood pressure measurements pre-, during and post-arterial compression.
In addition, digit Doppler and/or PVR measurements can be performed with the Falcon.
The PAT protocol includes schematic pictures, which help visualize to the examiner the required compression stage. The measured waveforms are clearly marked with automatic cursors to indicate when artery compression begins and when the compression is released. Thus, a quick visual assessment of the effect of compression on blood flow to the different parts of the hand is possible.
All sensors are color-coded, simplifying the interaction with the software. The ability to zoom in and out of the waveforms allows to quickly qualitatively determine whether a specific arterial compression has a patent collateral circulation or that harvesting the vessel will jeopardize the hand.
A decrease in the ipsilateral signal when compressing an artery, whether PPG waveform, blood pressure, PVR waveform, or Doppler spectrum, is indicative of impaired collateral circulation in the hand, and is generally considered as a contraindication to arterial harvesting.
For blood pressure assessment, a Finger Brachial Index (FBI) greater than 0.9 is considered normal. FBI is defined as the digit pressure divided by the ipsilateral brachial pressure. The determination of what is considered a significant drop in digit pressure varies in the literature but is in the range of a drop of 40 mmHg after radial/ulnar artery compression.
Example of a Palmar Arch Test specialty test performed on a patient using Viasonix Falcon/PRO. First, the Radial artery was compressed on two extreme digits, and then the Ulnar artery was compressed on the same digits.

Preoperative Assessment of Hand Circulation by Means of Doppler Ultrasonigraphy and the Modified Allen Test, Permyos Ruengsakulrach et al., J Thorac Cardiovasc Surg 2001;121:526-31
Assessment of collateral circulation to the hand prior to radial artery harvest, Joseph Habib, Laureen Baetz and Bhagwan Satiani, Vascular Medicine 17(5) 352– 361
Allen Test, Jonah Zisquit; James Velasquez; Nicholas Nedeff, National Center for Biotechnology Information, U.S. National Library of Medicine, NCBI Bookshelf, StatPearls Publishing LLC., 2020